Home
99. I2-Promoted Oxidative Annulation of Deoxybenzoin-Chalcone Adduct: Temperature-Controlled Access to Tetrasubstituted 2,3-trans-Dihydrofurans and Furans
Thi Thanh Nhan Vu, Thu Ngoc Trinh, Thu Trang Pham, Minh Tu Ha, Dinh Hung Mac,* Pascal Retailleau and Thanh Binh Nguyen*
Adv. Synth. Catal. 2024, 366,
DOI: 10.1002/adsc.202400304
Abstract: Tetrasubstituted 2,3-trans-dihydrofuran and furan are important heterocyclic scaffolds in natural product, bioorganic and medicinal chemistry as well as in material science. The synthesis of both of these heterocycles starting from common and readily available starting materials are challenging. We found that in situ generated deoxybenzoin-chalcone Michael adducts underwent oxidative annulation upon heating with molecular iodine in DMSO to provide selectively 2,3-trans-dihydrofurans at 80 °C and furans at 120 °C.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
98. Base- and Sulfur- Promoted Oxidative Lactonization of Chalcone-Acetate Michael Adducts: Access to Pyran-2-ones
Cao Nguyen Nguyen, Duc Toan Nguyen, Ha An Tran, Hung Dinh Mac,* Thi Thu Tram Nguyen, Pascal Retailleau and Thanh Binh Nguyen*
Org. Biomol. Chem. 2024, 22,
DOI: 10.1039/D4OB00479E
Abstract: A cost-effective, practical, straightforward and scalable synthesis of α-pyrones via base- and sulfur-promoted annulation of phenylacetates and chalcones. Generated in situ from the starting components by using dbu as a base catalyst, the Michael adducts underwent a smooth oxidative cyclization into 3,4,6-triaryl-2-pyranones upon heating with dabco and sulfur in DMSO. Extension to malonate in place of phenylacetates led to 4,6-diaryl-2-pyranone-2-carboxylates.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
97. Base-Catalyzed Synthesis of N-Aryl Thioacetamides from Multicomponent Reaction of Phenylacetylenes, Sulfur and Anilines
Minh Hang Nguyen, Hai Sam Nguyen, Le Anh Nguyen, Bich Ngoc Nguyen, Hai Thuong Cao, Dinh Hung Mac* and Thanh Binh Nguyen*
Eur. J. Org. Chem. 2024, 10.1002/ejoc.202301319
Abstract: In the presence of a
base catalyst (20 mol %) such as DBU, DABCO or Na2S•3H2O/N-methylpyrrolidone, phenylacetylenes
were found to efficiently undergo sulfurative amination with elemental sulfur
and anilines under neat conditions, leading to N-aryl thioamides with complete atom efficiency.
96. Sodium Sulfide-Promoted Regio-Defined Redox Condensation of o-Nitroanilines with Aryl Ketones to Benzo[a]phenazines and Quinoxalines
Duc Long Le, Le Anh Nguyen,* Ngoc Binh Vo, Thi Thu Tram Nguyen, Quoc Anh Ngo,* Pascal Retailleau and Thanh Binh Nguyen*
Org. Biomol. Chem. 2024, 22, 1167.
Abstract. Inexpensive sodium sulfide trihydrate was found to promote unprecedented 6e- regio-predefined redox condensation of o-nitroanilines with α-tetralones to benzo[a]phenazines. The method was also successfully extended to acetophenones and higher homologs as the reducing partners to provide 2-phenylquinoxalines. Compared to traditional approaches to benzo[a]phenazine and quinoxaline cores starting with o-phenylenediamines, the present strategy could afford these heterocycles with well-defined regiochemistry based on the structure of starting o-nitroanilines.
95. Sulfur- and Amine- Promoted Multielectron Autoredox Transformation of Nitromethane: Multicomponent Access to Thiourea Derivatives
Supasorn Phaenok, Le Anh Nguyen, Darunee Soorukram, Thi Thanh Tam Nguyen, Pascal Retailleau, Thanh Binh Nguyen*
Chem. Eur. J. 2023, 10.1002/chem.202303703
Abstract. Thiourea derivatives are in-demand motifs in organic synthesis, medicinal chemistry and material science, yet redox methods for the synthesis that start from safe, simple, inexpensive and readily available feedstocks are scarce. In this article, we disclose the synthesis of these motifs using elemental sulfur and nitromethane as the starting materials. The method harnesses the multi-electron auto-redox property of nitromethane in the presence of sulfur and amines, delivering thiourea products without any added oxidant or reductant. Extension of this reaction to cyclizable amines and/or higher homologues of nitromethane led to a wide range of nitrogen heterocycles and thioamides. Operationally simple, the reactions are scalable, tolerate a wide range of functional groups, and can be employed for the direct functionalization of natural products. Mechanistically, the nitro group was found to act as an oxidant leaving group, being reduced to ammonia whereas sulfur, along with the role of a sulfur building block for the thiocarbonyl group, behaved as a complementary reductant, being oxidized to sulfate.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
94. DABCO-Catalyzed DMSO-Promoted Sulfurative 1,2-Diamination of Phenylacetylenes with Elemental Sulfur and o-Phenylenediamines: Access to Quinoxaline-2-thiones
Thi Minh Chau Tran, Nang Duy Lai, Thai Thanh Thu Bui, Dinh Hung Mac*, Thi Thu Tram Nguyen, Pascal Retailleau, and Thanh Binh Nguyen*
Org. Lett. 2023, 25, 7225-7229.
Abstract. The oxidative amination of alkynes typically requires transition metal catalysts and strong oxidants. Herein, we alternatively utilize DABCO as a sulfur-activating catalyst to achieve the sulfurative 1,2-diamination of phenylacetylenes with elemental sulfur and o-phenylenediamines. DMSO was found to be particularly suitable for use as a terminal oxidant for this three-component process. A mechanistic study has shown that this cascade reaction is triggered by the addition of active sulfur species to the triple bond of phenylacetylenes.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
93. Sulfur-Promoted Oxidative Cyclization of Pentan-1-ones: Direct Access to Tetrasubstituted Furans from Deoxybenzoins and Chalcones
Van Phu Nguyen, Nhu Ngan Ha Nguyen, Nang Duy Lai, Dinh Hung Mac*, Pascal Retailleau, and Thanh Binh Nguyen*
Org. Lett. 2023, 25, 6419-6322.
Abstract. Furan is an important heterocyclic scaffold in natural product, bioorganic, and medicinal chemistry as well as in materials science. The system S8/DABCO/DMSO was found to efficiently mediate the oxidative cyclization of 1,2,3,5-tetraarylpentan-1-ones A, which were obtained in situ as the Michael adducts of chalcones 1 and deoxybenzoins 2, to furan 3. The strategy provided convenient and direct access to tetrasubstituted furans 3 from readily available starting materials with high functional group tolerance.
92. Synthesis of Thioureas, Thioamides, and Aza-Heterocycles via Dimethyl-Sulfoxide-Promoted Oxidative Condensation of Sulfur, Malonic Acids, and Amines
Trung Hieu Do, Supasorn Phaenok, Darunee Soorukram, Tina Modjinou, Daniel Grande, Thi Thanh Tam Nguyen*, and Thanh Binh Nguyen*
Org. Lett. 2023, 25, 6322-6327.
Abstract. Malonic acid and derivatives have been well-known to undergo monodecarboxylation under relatively mild conditions and have been exclusively used as a C2 synthon. We report herein their new application as a C1 synthon via double decarboxylation promoted by sulfur and dimethyl sulfoxide. In the presence of amines as nucleophiles, a wide range of thioureas and thioamides as well as N-heterocycles were obtained in good to excellent yields under mild heating conditions.
Le Anh Nguyen, Supasorn Phaenok, Duc Long Le, Thi Thu Tram Nguyen, Quoc Anh Ngo*, and Thanh Binh Nguyen*
Org. Lett. 2023, 25, 5145-5150.
Abstract. As frequently encountered byproducts of isocyanate chemistry, hydrogen sulfide and related sulfur containing compounds should be treated in a safe way to lower their adverse health and environmental effects, especially in large scale syntheses. As a proof of concept, we report herein an example of in situ recycling of sulfur byproduct to reductant in the synthesis of bioactive 2-aminobenzoxazoles 3. Using an Fe/S catalytic system, this heterocyclic scaffold could be obtained from o-nitrophenols 1 with isothiocyates 2 via direct redox condensation consisting of reduction of the nitro group of 1 by the sulfur moiety of 2.
90. Sulfur-Promoted Oxidative Condensation of Chalcones with Unsubstituted Cyanoacetamide in DMSO: Access to 3-Cyanopyrid-2-ones
Bich Ngoc Nguyen, Minh Hieu Tran, Thai Thanh Thu Bui, Dinh Hung Mac*, Van Phong Pham*, Pascal Retailleau, and Thanh Binh Nguyen*
J. Org. Chem. 2023, 88, 15, 11197–11204
Abstract.
Elemental sulfur and DABCO were found to be an excellent combination to
promote a one-pot cascade of condensation-oxidative cyclization of
chalcones and unsubstituted cyanoacetamide in DMSO to provide
3-cyanopyrid-2-ones.
89. Reaction of 1-acetonaphthones with anilines and elemental sulfur: rapid construction of 1-anilinonaphtho[2,1-b]thiophenes
Hoang Yen Nguyen, Thi Minh Chau Tran, Van Ha Nguyen, Pascal Retailleau, Dinh Hung Mac* and Thanh Binh Nguyen*
Org. Biomol. Chem. 2023, 21, 503-507.
Abstract. 1-Anilinonaphtho[2,1-b]thiophenes could be conveniently synthesized from a three-component reaction of 1-acetonaphthones with anilines and elemental sulfur under catalyst-free simple heating conditions.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
88. Direct access to thieno[3,4-b]thiophenes via elemental sulfur-promoted sulfurative tetramerization of acetophenones
Thanh Binh Nguyen* and Pascal Retailleau
Chem. Commun. 2022, 58, 13333-13336.
Abstract. Thieno[3,4-b]thiophene cores could be conveniently obtained via one-pot sulfurative tetramerization of acetophenones using a ternary system comprised of elemental sulfur, a base and DMSO under simple heating conditions. The structure of the products was verified by X-ray crystallography.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
87. Sulfur–DMSO promoted oxidative coupling of active methylhetarenes with amines: access to amides
Thi Thu Tram Nguyen, Viet Dung Duong,Thi Ngoc Nga Pham, Quoc Thanh Duong and Thanh Binh Nguyen*
Org. Biomol. Chem. 2022, 20, 8054-8058.
Abstract.The elemental sulfur–DMSO couple was found to efficiently promote the oxidative coupling of active methylhetarenes with amines to yield amides under simple heating conditions. When 2-methylquinoline was used as the methylhetarene component, the formation of the expected 2-quinolinecarboxamides from anilines could be efficiently catalyzed by iron, nickel and cobalt salts. The method displayed good functional group tolerance and was applicable to aromatic, heteroaromatic and aliphatic amines. Other substrates such as phenylacetic acid, dibenzyl disulfide, and benzylamine could act as competent partners in place of methylhetarenes.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
86. DABCO-Catalyzed Reaction of 2‑Naphthols with Aryl Isothiocyanates: Access to 2‑Iminonaphtho-1,3-oxathioles
Thanh Binh Nguyen,* and Pascal Retailleau
Org. Lett. 2022, 23, 6676–6680.
DOI:10.1021/acs.orglett.2c02736
Abstract.2-Naphthols 1were found to react with aryl isothiocyanates 2 to provide 2-iminonaphtho-1,3-oxathioles 3 in the presence of DABCO as a catalyst. When elemental sulfur was used, moderate to good yields of 3 could be achieved from a nearly equimolar mixture of two starting materials 1 and 2. Products 3 could also be formed in the absence of sulfur or an external oxidant. In the latter case, an additional equivalent of 2 was found to act as a dehydrogenating agent.
85. Base-Catalyzed Multicomponent Access to Quinoxalin-2-thiones from o-Phenylenediamines, Aryl Ketones and Sulfur
Thanh Binh Nguyen,* Hung Dinh Mac,* Thi Minh Chau Tran, Bich Ngoc Nguyen and Hai Thuong Cao
Org. Biomol. Chem. 2022, 20, 7226.
DOI: 10.1039/D2OB01343F
Abstract.3-Arylquinoxaline-2-thiones were conveniently synthesized via three-component oxidative condensation of acetophenones with o-phenylenediamines and sulfur in DMSO in the presence of piperidine as a catalyst. The products could be readily isolated from the reaction mixture by simple precipitation and washing with methanol. This set of reaction conditions applied to higher homologs of acetophenones as well as benzyl phenyl ketones led to 2,3-di-C-substituted quinoxalines. Further functionalization of 3-phenylquinoxaline-2-thione via reaction on the thione group could be readily performed to provide quinoxaline derivatives in good yields.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
84. Sulfur-Catalyzed Oxidative Condensation of Aryl Alkyl Ketones with o-Phenylenediamines: Access to Quinoxalines
Le Anh Nguyen, Thi Thu Tram Nguyen, Quoc Anh Ngo,* Thanh Binh Nguyen*
Adv. Synth. Catal. 2022, 364, 2748-2752.
DOI: 10.1002/adsc.202200527
Nguyen, L. A.; Nguyen, T. T. T.; Ngo, Q. A.; Nguyen, T. B. Adv. Synth. Catal. 2022, 364, 2748-2752.
Abstract. While elemental sulfur has been largely used as oxidant or sulfurating agent, its role as a catalyst has not been developed. We report here its catalytic activity in the oxidative condensation of o-phenylenediamines with acetophenones in DMSO to provide quinoxalines. The method was also extended to α-tetralones, propiophenones (R=Me) as well as higher homologues (R=Et, n-Pr) in place of acetophenones, leading to a wide range of naphthoquinoxalines and 3-substituted 2-arylquinoxalines.
83. Direct access to 2-aryl-3-cyanothiophenes by a base-catalyzed one-pot two-step three-component reaction of chalcones with benzoylacetonitriles and elemental sulfur
Nang Duy Lai, Thu Trang Nguyen, Nhu Ngan Ha Nguyen, Pascal Retailleau, Dinh Hung Mac* and Thanh Binh Nguyen*
DOI: 10.1039/D2QO00526C
Org. Chem. Front. 2022, 9, 3163-3168.Abstract. 3-Cyanothiophene is an important heterocyclic scaffold in bioorganic and
medicinal chemistry as a useful synthetic intermediate as well as in
materials science as a privileged motif for photovoltaic development.
Herein, we report our unexpected results on the formation of
3-cyanothiophene derivatives as the major products via a
three-component reaction of chalcones, benzoylacetonitriles and
elemental sulfur along with the minor products 2-aminothiophenes. The
ratios between these two thiophene products is 4 : 3 and could be varied
by simply changing the promoting base as well as its stoichiometric
ratio. The method was successfully extended to benzoylacetate in place
of benzoylacetonitrile to provide thiophene-3-carboxylates.
82. Convenient Synthesis of Triphenylphosphine Sulfide from Sulfur and Triphenylphosphine
Thanh Binh Nguyen*
Clean Technol. 2022, 4, 234-238. Open access
Nguyen, T. B. Clean Technol. 2022, 4, 234-238.
DOI: 10.3390/cleantechnol4020013
Abstract. Elemental sulfur (S8) was found to react very rapidly (<1
min) with a stoichiometric amount of triphenylphosphine at rt in
sufficient amount of solvent (0.2–0.5 mL of solvent/1 mmol of PPh3).
Compared to the previously described methods, the present procedure
constitute excellent access to triphenylphosphine sulfide.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
81.
TFA/DMSO-Promoted Cross-Dehydrogenative Coupling of Hetaryl Thiols with
Indoles: Access to 3-(Hetarylsulfenyl)indole under Mild Conditions
Tan Sang Truong, Pascal Retailleau, Thanh Binh Nguyen*
Asian J. Org. Chem. 2022,11, e202100751
DOI: 10.1002/ajoc.202100751
Truong, T. T.; Retailleau, P.; Nguyen, T. B. Asian J. Org. Chem. 2022, 11, e202100751Abstract.
TFA/DMSO combination was found to efficiently promote the
cross-dehydrogenative coupling of a wide range of hetaryl thiols with
indole derivatives, leading to 3-(hetarylsulfenyl)indoles in good-to
excellent yields under mild heating conditions. Two-fold
hetarylsulfenylation of indoles in 2- and 3- positions could be
conveniently achieved by simply adapting molar ratio of the starting
materials.
80. Base-Catalyzed Three-Component Reaction between Chalcones, Isothiocyanates and Sulfur: Access to Thiazole-2-thiones
Thanh Binh Nguyen,* and Pascal Retailleau
Org. Lett. 2021, 23, 5344-5348.
DOI:10.1021/acs.orglett.1c01653
Nguyen, T. B.; Retailleau, P. Org. Lett. 2021, 23, 5344-5348.
Abstract. A convenient synthesis of thiazole-2-thiones was developed based on base-catalyzed three-component reac-tions between chalcones, isothiocyanates and elemental sulfur.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
79. Base-Catalyzed Three-Component Reaction of α-Cyanoacetates with Chalcones and Elemental Sulfur: Access to 2-Aminothiophenes Unobtainable via the Gewald Reaction
Thanh Binh Nguyen,* Dinh Hung Mac,* and Pascal Retailleau
J. Org. Chem. 2021, 86, 9418-9427.
DOI: 10.1021/acs.joc.1c00740
Nguyen, T. B.; Mac, D. H.; Retailleau, P. J. Org. Chem. 2021, 86, 9418-9427.
Abstract. While Gewald reaction was well known for more than haft a century as an excellent method providing bioactive 2-aminothiophenes from reactions of α-cyanoacetates and carbonyl compounds and elemental sulfur, its application to dibenzoylmethanes as the carbonyl substrates was however unknown and experimentally proven unsuccessful. We proposed here a convenient approach to such series of compounds by a DABCO catalyzed, one-pot, two-step, three-component reaction of α-cyanoacetate with chalcones and elemental sulfur. This catalytic strategy is highlighted by its excellent atom/step efficiency and high degree of structural diversification by simply choosing the suitable starting chalcones, which are unarguably much more readily available than dibenzoylmethanes.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
78. Fe/S-Catalyzed synthesis of 2-benzoylbenzoxazoles and 2-quinolylbenzoxazoles via redox condensation of o-nitrophenols with acetophenones and methylquinolines
Le Anh Nguyen, Thi Thu Tram Nguyen, Quoc Anh Ngo,* Thanh Binh Nguyen*
Org. Biomol. Chem. 2021, 19, 6015-6020.
DOI:10.1039/D1OB00976A
Nguyen, L. A.; Nguyen, T. T. T.; Ngo, Q. A.; Nguyen, T. B. Org. Biomol. Chem. 2021, 19, 6015-6020.
Abstract. An Fe/S catalyst generated in situ from FeCl2·4H2O and elemental sulfur S8 in the presence of a tertiary amine as a base was found to catalyze efficiently a 6e− redox condensation of o-nitrophenols
with acetophenones and methylquinolines. The condensed products
2-benzoylbenzoxazoles and 2-quinolylbenzoxazoles were obtained in
reasonable yields with water as the only byproduct at a temperature as
low as 80 °C.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
77. Break-and-Build Strategy for the Synthesis of 2-Benzoylbenzoxazoles from o-Aminophenols and Acetophenones
Le Anh Nguyen, Quoc Anh Ngo,* Pascal Retailleau, Thanh Binh Nguyen*
Adv. Synth. Catal. 2021, 363, 2098-2103.
DOI:10.1002/adsc.202100150
Nguyen, L. A.; Ngo, Q. A.; Retailleau, P.; Nguyen, T. B. Adv. Synth. Catal. 2021, 363, 2098.
Abstract. Although compounds with a 2-benzoylbenzoxazole motif are biologically relevant, there are only a few methods for synthesizing them, most of which relied on multistep process or required substrates bearing activating groups. Herein, we report an efficient method for the synthesis of such compounds by direct reactions of o-aminophenols with acetophenones promoted by sulfur in DMSO. The reaction was found to proceed via a Willgerodt rearrangement-type benzoxazolation of acetophenones followed by a benzylic oxidation to reinstall the carbonyl function. This method has a broad substrate scope and good tolerance for sensitive functional groups.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
76. The catalytic role of elemental sulfur in the DMSO-promoted oxidative coupling of methylhetarenes with amines: synthesis of thioamides and bis-aza-heterocycles
Thi Thu Tram Nguyen, Le Anh Nguyen, Quoc Anh Ngo,* Marina Koleski and Thanh Binh Nguyen*
Org. Chem. Front. 2021, 8, 1593.
DOI: 10.1039/D0QO01654C
Nguyen, T. T. T.; Nguyen, L. A.; Ngo, Q. A.; Koleski, M.; Nguyen, T. B. Org. Chem. Front. 2021, 8, 1593-1598.
Abstract. Thioamides could be conveniently synthesized in good to excellent yields via DMSO-promoted oxidative coupling of methylhetarenes with amines in the presence of a near stoichiometric amount of sulfur (1.25 equiv.). Both aliphatic and aromatic amines were found to be competent substrates. When anilines o-substituted by cyclizable groups such as OH, NH2, NHPh, SH and CONH2 were used as amine substrates, the corresponding hybrid bis-aza-heterocycles were formed in high yields even with a sulfur loading as low as 0.5 equiv.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
75. Room-Temperature Synthesis of Tetrasubstituted 1,3-Dithioles by Dimerizing Sulfuration of Chalcones with Elemental Sulfur
Thanh Binh Nguyen,* Dinh Hung Mac,* and Pascal Retailleau
J. Org. Chem. 2020, 85, 13508-13516.
DOI: 10.1021/acs.joc.0c01618
Nguyen, T. B.; Mac, D. H.; Retailleau, P. J. Org. Chem. 2020, 85, 13508.
Abstract. Chalcones were found to undergo sulfurative dimerization with elemental
sulfur to tetrasubstituted 1,3-dithioles. The reaction was found to
proceed at room temperature in the presence of a nitrogen-base catalyst
in DMSO.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
74. Umpolung Strategy for Oxidative Functionalization with 2-Aminothiophenols: Access to Complex 2-Arylbenzothiazine Derivatives from Simple Alkyl Aryl Ketones
Thanh Binh Nguyen,* and Pascal Retailleau
Adv. Synth. Catal. 2020, 362, 5380-5384.
DOI: 10.1002/adsc.202000964
Nguyen, T. B.; Retailleau, P. Adv. Synth. Catal. 2020, 362, 5380.
Abstract. Strong Brønsted acids were found to catalyze the oxidative condensation of 2-aminothiophenols with aryl alkyl ketones in DMSO to provide a wide range of complex 2-arylbenzothiazines. With acetophenones, dimeric 2-arylbenzothiazines were formed. On the other hand, the reaction with propiophenones resulted in 2:1 adducts whereas 1:1 adducts were produced with isobutyrophenones, benzoylacetonitrile and ethyl benzoylacetate.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
73. Access to [2,1]Benzothiazine S,S-Dioxides from β-Substituted o-Nitrostyrenes and Sulfur
Thi Mo Nguyen, Hoang Anh Cao, Thi Thuong Thuong Cao, Satoki Koyama, Dinh Hung Mac*, and Thanh Binh Nguyen*J. Org. Chem. 2020, 85, 12058.
Nguyen, T. M.; Cao, H. A.; Cao, T. T. T.; Koyama, S.; Mac, D. H.; Nguyen, T. B. J. Org. Chem. 2020, 85, 12058.
Abstract. [2,1]Benzothiazine S,S-dioxides 2 were synthesized by simply heating o-nitrostyrenes
with elemental sulfur in 3-picoline with complete atom economy. This
reaction was found to occur without any added catalyst and consist of a
cascade of reduction of the nitro group, sulfuration of a C–H of the
double bond, oxidation of a sulfur atom to its highest oxidation state
by the migration of two oxygen atoms from the nitro group, and formation
of new N–S bonds. Furthermore, the method could also be applied to o-nitrocinnamamides and cinnamate esters.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
72. Umpolung Strategy for α,α’‐Functionalization of Ketones with 2‐Aminothiophenols: Stereoselective Access to Spirobis(1,4‐Benzothiazines)
Thanh Binh Nguyen,* and Pascal RetailleauAdv. Synth. Catal. 2020, 362, 3824.
DOI: 10.1002/adsc.202000725
Nguyen, T. B.; Retailleau, P. Adv. Synth. Catal. 2020, 362, 3824.
Abstract. In the presence of TFA as a strong acid catalyst in DMSO, α,α’‐enolizable ketones were found to be stereoselectively α,α’‐functionalized with 2‐aminothiophenols to provide spirobis(1,4‐benzothiazine) derivatives.
Adv. Synth. Catal. 2020, 362, 3448.
DOI: 10.1002/adsc.202000535
Abstract. In the presence of TFA as a strong acid catalyst in DMSO, α,α’‐enolizable ketones were found to be stereoselectively α,α’‐functionalized with 2‐aminothiophenols to provide spirobis(1,4‐benzothiazine) derivatives.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
70. Recent Advances in the Synthesis of Heterocycles via Reactions Involving Elemental Sulfur
Thanh Binh Nguyen*Adv. Synth. Catal. 2020, 362, 3448.
DOI: 10.1002/adsc.202000535
Nguyen, T. B. Adv. Synth. Catal. 2020, 362, 3448.
Abstract. It is well known that heterocycles are among the most significant molecules for everyday life, ranging from natural products and bioactive substances to functional materials. This review will focus on the synthesis of heterocycles by reactions involving elemental sulfur published from 2017 until now.
Eur. J. Org. Chem. 2020, 3818.
DOI: 10.1002/ejoc.202000523
Abstract. It is well known that heterocycles are among the most significant molecules for everyday life, ranging from natural products and bioactive substances to functional materials. This review will focus on the synthesis of heterocycles by reactions involving elemental sulfur published from 2017 until now.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
69. Sulfur‐Promoted Synthesis of Benzoxazoles from 2‐Aminophenols and Aldehydes
Le Anh Nguyen, Thai Duy Dang, Quoc Anh Ngo,* Thanh Binh Nguyen*Eur. J. Org. Chem. 2020, 3818.
DOI: 10.1002/ejoc.202000523
Nguyen, L. A.; Dang, T. D.; Ngo, Q. A.; Nguyen, T. B. Eur. J. Org. Chem. 2020, 3818.
Abstract. Oxidation with sulfur: Elemental sulfur (S8) was found to be an excellent stoichiometric oxidant to promote oxidative condensation of 2‐aminophenols with a wide range of aldehydes to provide benzoxazoles.
Chem. Eur. J. 2020, 26, 4682
DOI: 10.1002/chem.201905597
Nguyen, T. B.; Nguyen, T. M.; Retailleau, P. Chem. Eur. J. 2020, 26, 4682.
Abstract. In this study we reported on a very simple technique to perform efficiently photodimerization of some vinylpyridines. By irradiating a stirred mixture of several stilbazoles with solid oxalic acid dihydrate dispersed in a non‐polar (e. g. cyclohexane) or moderately polar (benzene, dichloromethane, dioxane) solvent, the corresponding dimeric cyclobutane adducts were obtained in high yields with excellent of regio‐ and stereo‐ selectivities. The strategy could also be applied successfully to oily, waxy or even insoluble stilbazoles. Moreover, the oxalic acid loading could be lowered to sub‐stoichiometric amounts. When further optimizations were needed, our strategy was found to be highly flexible to identify other oligocarboxylic acids as alternative additive to improve or even overturn regioselectivity. Oxalic acid and other oligocarboxylic acids were found to be capable of orienting more than fifty stilbazoles toward photodimerization under these conditions.
Adv. Synth. Catal. 2020, 362, 160.
DOI: 10.1002/adsc.201901235
Nguyen, T. T. T.; Le, V. A.; Retailleau P.; Nguyen, T. B. Adv. Synth. Catal. 2020, 362, 160.
Abstract. A straightforward access to 2‐amino‐3‐arylthiophenes has been developed via one‐pot two‐step three‐component reaction of arylacetonitriles, chalcones and elemental sulfur. The first step consists of a DBU‐catalyzed formation of Michael adduct between arylacetonitriles and chalcones. The second step is a cascade of DABCO‐catalyzed sulfuration of the Michael adduct with elemental sulfur followed by an oxidative cyclization to afford thiophenes. Compared to the Gewald reactions and related transformations which are limited in acetonitriles bearing a methylene group activated by an α‐substituted electron withdrawing group as substrates, our method can be applied to a wide range of arylacetonitriles and requires only catalytic amounts of DBU and DABCO. The developed reaction opens an access to 3‐aryl‐2‐aminothiophenes complementary to classical Gewald reactions with high degree of structural diversity and atom efficiency.
Org. Lett. 2019, 21, 6570
DOI: 10.1021/acs.orglett.9b02558.
Nguyen, T. B.; Nguyen, L. A.; Retailleau P. Org. Lett. 2019, 21, 6570.
Abstract. The S8/DMSO combination was found to be an excellent oxidizing agent for ipso,α,β,γ-tetramination of cyclohexanones with two molecules of o-phenylenediamines in the presence of TFA as a catalyst. The strategy could be applied to pyrido cyclohexanes as a direct method to extend the fused heteroaromatic system.
Org. Biomol. Chem. 2019, 17, 6355.
Abstract. An efficient synthesis of 2H-3-nitrothiochromenes via a cascade reaction was established. Starting from commercially available o-bromobenzaldehydes and β-nitrostyrenes with sodium sulfide nonahydrate as an inexpensive sulfur source, various substituted thiochromenes were synthesized with high functional group tolerance without any added transition metal catalyst or additive.
Adv. Synth. Catal. 2019, 361, 3337.
DOI: 10.1002/adsc.201900371
Nguyen, T. B.; Retailleau P. Adv. Synth. Catal. 2019, 10.1002/adsc.201900371
Abstract. A sulfur-promoted three-component reaction of isatoic anhydride, primary aliphatic or aromatic amines, and acetophenones leading to densely substituted 3-substituted 2-aroylquinazolin-4(3H)-ones is reported. The key step involves a cascade reaction of selective oxidation of the methyl group of the acetophenones, followed by a condensation with anthranilamides. The scope of the reaction is applicable to the synthesis of tryptanthrin and various 3-unsubstituted 2-aroylquinazolin-4(3H)-ones.
Adv. Synth. Catal. 2019, 361, 3588.
DOI: 10.1002/adsc.201900436
Nguyen, T. B.; Retailleau P. Adv. Synth. Catal. 2019, 10.1002/adsc.201900436
Abstract. Oxidation with sulfur: Elemental sulfur (S8) was found to be an excellent stoichiometric oxidant to promote oxidative condensation of 2‐aminophenols with a wide range of aldehydes to provide benzoxazoles.
68. [2+2]‐Photodimerization of Stilbazoles Promoted by Oxalic Acid in Suspension
Thanh Binh Nguyen,* Tuan Minh Nguyen, Pascal RetailleauChem. Eur. J. 2020, 26, 4682
DOI: 10.1002/chem.201905597
Nguyen, T. B.; Nguyen, T. M.; Retailleau, P. Chem. Eur. J. 2020, 26, 4682.
Abstract. In this study we reported on a very simple technique to perform efficiently photodimerization of some vinylpyridines. By irradiating a stirred mixture of several stilbazoles with solid oxalic acid dihydrate dispersed in a non‐polar (e. g. cyclohexane) or moderately polar (benzene, dichloromethane, dioxane) solvent, the corresponding dimeric cyclobutane adducts were obtained in high yields with excellent of regio‐ and stereo‐ selectivities. The strategy could also be applied successfully to oily, waxy or even insoluble stilbazoles. Moreover, the oxalic acid loading could be lowered to sub‐stoichiometric amounts. When further optimizations were needed, our strategy was found to be highly flexible to identify other oligocarboxylic acids as alternative additive to improve or even overturn regioselectivity. Oxalic acid and other oligocarboxylic acids were found to be capable of orienting more than fifty stilbazoles toward photodimerization under these conditions.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
67. Access to 2‐Amino‐3‐Arylthiophenes by Base‐Catalyzed Redox Condensation Reaction Between Arylacetonitriles, Chalcones, and Elemental Sulfur
Thi Thu Tram Nguyen, Van Anh Le, Pascal Retailleau, and Thanh Binh Nguyen*Adv. Synth. Catal. 2020, 362, 160.
DOI: 10.1002/adsc.201901235
Nguyen, T. T. T.; Le, V. A.; Retailleau P.; Nguyen, T. B. Adv. Synth. Catal. 2020, 362, 160.
Abstract. A straightforward access to 2‐amino‐3‐arylthiophenes has been developed via one‐pot two‐step three‐component reaction of arylacetonitriles, chalcones and elemental sulfur. The first step consists of a DBU‐catalyzed formation of Michael adduct between arylacetonitriles and chalcones. The second step is a cascade of DABCO‐catalyzed sulfuration of the Michael adduct with elemental sulfur followed by an oxidative cyclization to afford thiophenes. Compared to the Gewald reactions and related transformations which are limited in acetonitriles bearing a methylene group activated by an α‐substituted electron withdrawing group as substrates, our method can be applied to a wide range of arylacetonitriles and requires only catalytic amounts of DBU and DABCO. The developed reaction opens an access to 3‐aryl‐2‐aminothiophenes complementary to classical Gewald reactions with high degree of structural diversity and atom efficiency.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
66. Strategy for Contiguous Tetramination of Cyclohexanones with o-Phenylenediamines with Elemental Sulfur and DMSO
Thanh Binh Nguyen,* Le Anh Nguyen, and Pascal RetailleauOrg. Lett. 2019, 21, 6570
DOI: 10.1021/acs.orglett.9b02558.
Nguyen, T. B.; Nguyen, L. A.; Retailleau P. Org. Lett. 2019, 21, 6570.
Abstract. The S8/DMSO combination was found to be an excellent oxidizing agent for ipso,α,β,γ-tetramination of cyclohexanones with two molecules of o-phenylenediamines in the presence of TFA as a catalyst. The strategy could be applied to pyrido cyclohexanes as a direct method to extend the fused heteroaromatic system.
65. Convenient one-pot access to 2H-3-nitro-thiochromenes from 2-bromobenzaldehydes, sodium sulfide and β-nitrostyrenes
Thi Thu Huong, Le, Chitose Youhei, Thanh Binh Nguyen* and Dinh Hung Mac*Org. Biomol. Chem. 2019, 17, 6355.
Abstract. An efficient synthesis of 2H-3-nitrothiochromenes via a cascade reaction was established. Starting from commercially available o-bromobenzaldehydes and β-nitrostyrenes with sodium sulfide nonahydrate as an inexpensive sulfur source, various substituted thiochromenes were synthesized with high functional group tolerance without any added transition metal catalyst or additive.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
64. Sulfur-Promoted Synthesis of 2-Aroylquinazolin-4(3H)-ones by Oxidative Condensation of Anthranilamide and Acetophenones
Thanh Binh Nguyen,* Hou Jing-ya, and Pascal RetailleauAdv. Synth. Catal. 2019, 361, 3337.
DOI: 10.1002/adsc.201900371
Nguyen, T. B.; Retailleau P. Adv. Synth. Catal. 2019, 10.1002/adsc.201900371
Abstract. A sulfur-promoted three-component reaction of isatoic anhydride, primary aliphatic or aromatic amines, and acetophenones leading to densely substituted 3-substituted 2-aroylquinazolin-4(3H)-ones is reported. The key step involves a cascade reaction of selective oxidation of the methyl group of the acetophenones, followed by a condensation with anthranilamides. The scope of the reaction is applicable to the synthesis of tryptanthrin and various 3-unsubstituted 2-aroylquinazolin-4(3H)-ones.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
63. Sulfur‐Catalyzed Stereo and Regioselective Synthesis of Heteropropellanes via Oxidative Condensation of Cyclohexanones with 2‐Aminophenols
Thanh Binh Nguyen* and Pascal Retailleau
Adv. Synth. Catal. 2019, 361, 3588.
DOI: 10.1002/adsc.201900436
Nguyen, T. B.; Retailleau P. Adv. Synth. Catal. 2019, 10.1002/adsc.201900436
Abstract. Sulfur was found to be an excellent catalyst for the oxidative coupling of cyclohexanones with o-aminophenols using DMSO as oxidant. A wide range of heteropropellanes were obtained with complete regio and stereoselectivities and functional group tolerance.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
62. Sulfur-Promoted DABCO-Catalyzed Oxidative Trimerization of Phenylacetonitriles
Thanh Binh Nguyen* and Pascal RetailleauJ. Org. Chem. 2019, 84, 5907.
DOI: 10.1021/acs.joc.9b00408
Nguyen, T. B.; Retailleau P. J. Org. Chem. 2019, DOI: 10.1021/acs.joc.9b00408
Abstract. By simply heating phenylacetonitriles 1 with elemental sulfur and DMSO in the presence of a catalytic amount of DABCO, we have performed an oxidative trimerization leading to polysubstituted pyrrole heterocycles 2 in excellent yields and E-configuration.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
61. Elemental Sulfur/DMSO‐Promoted Multicomponent One‐pot Synthesis of Malonic Acid Derivatives from Maleic Anhydride and Amines
Le Anh Nguyen, Pascal Retailleau and Thanh Binh Nguyen*Adv. Synth. Catal. 2019, 361, 2864.
DOI: 10.1002/adsc.201900160
Nguyen, L. A.; Retailleau P.; Nguyen, T. B. Adv. Synth. Catal. 2019, DOI: 10.1002/adsc.201900160
Abstract. Malonic acid derivatives could be conveniently prepared with high degree of functional flexibility via redox condensation reactions between anhydride maleic, amines, elemental sulfur and DMSO as oxidant. This multicomponent decarboxylative transformation consists in a cascade of ring opening, decarboxylative oxidative thioamidation at temperature as low as 50 °C.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
60. Sulfur‐Catalyzed Oxidative Coupling of Dibenzyl Disulfides with Amines: Access to Thioamides and Aza Heterocycles
Thanh Binh Nguyen,* Le Phuong Anh Nguyen and Thi Thu Tram NguyenAdv. Synth. Catal. 2019, 361, 1787.
DOI: 10.1002/adsc.201801695
Nguyen, T. B.; Nguyen, L. P. A.; Nguyen, T. T. T. Adv. Synth. Catal. 2019, DOI: 10.1002/adsc.201801695
Abstract. In the presence of catalytic amounts of elemental sulfur, dibenzyl disulfide/DMSO was found to be an excellent thiobenzoylating agent of amines to provide a wide range of thioamides. The reaction becomes autocatalytic when anilines substituted by an o‐cyclizable group were used as nucleophile, leading to the corresponding 2‐aryl aza heterocycles.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
59. Sulfur-Promoted Decarboxylative Sulfurative Hexamerization of Phenylacetic Acids: Direct Approach to Hexabenzylidyne TetrasulfidesThanh Binh Nguyen* and Pascal Retailleau
Org. Lett. 2019, 21, 279.
DOI: 10.1021/acs.orglett.8b03728
Nguyen, T. B.; Retailleau, P. Org. Lett. 2019, 21, 279.
Abstract. During our experiments aimed at understanding the reaction pathways by which arylacetic acids were oxidatively decarboxylated and condensed with different nucleophiles in the presence of elemental sulfur, these acids have been treated with sulfur powder in dimethyl sulfoxide (DMSO) and N-methylpiperidine in the absence of nucleophiles, producing a remarkable symmetrical sulfurated hexamer consisting of six benzylidyne moieties and four sulfur atoms.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
58. Sulfur-Promoted Aminative Aromatization of 1,2,3,4- Tetrahydrophenazines with Amines: Flexible Access to 1- AminophenazinesThanh Binh Nguyen* and Pascal Retailleau
Adv. Synth. Catal. 2018, 360, 2389.
DOI: 10.1002/adsc.201800260
Nguyen, T. B.; Retailleau, P. Adv. Synth. Catal. 2018, 360, 2389.
Abstract. Elemental sulfur was found to be an excellent reagent for promoting the aminative aromatization of 1,2,3,4-tetrahydrophenazines with amines to provide a wide range of functionalized 1-aminophenazines.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
57. Unaromatized Tetrahydrobenzimidazole Synthesis from p-Benzoquinone and N-Arylamidines and their Cytotoxic Potential
M. Q. Tran, T. B. Nguyen, W. R. Sawadogo, L. Ermolenko, S. Song, P. Retailleau, M. Diederich, A. Al-Mourabit, Eur. J. Org. Chem. 2018, 5878.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
56. Toward the Synthesis of Sceptrin and Benzosceptrin: Solvent Effect in Stereo- and Regio- Selective [2 + 2] Photodimerization and easy Access to the Fully Substituted Benzobutene System
Thanh Binh Nguyen, Le Anh Nguyen, Mathilde Corbin, Pascal Retailleau, Ludmila Ermolenko, and Ali Al-Mourabit,
Eur. J. Org. Chem. 2018, 5861.
DOI: 10.1002/ejoc.201701607
Nguyen, T. B.; Nguyen, L. A.; Corbin, M.; Retailleau, P.; Ermolenko, L.; Al-Mourabit, A. Eur. J. Org. Chem. 2018, 5861.
Abstract. By simply switching solvents, two sets of conditions for total regio- and stereo- selective photodimerization of (E)-3-(imidazo[1,2-a]pyrimidin-2-yl)acrylic acid (E)-7a were found. In acetonitrile in the presence of benzophenone as a photosensitizer or in cyclohexane, head-to-head cis-trans-cis isomer 8a was the only observable cycloadduct. In DMSO, head-to-head trans-trans-trans isomer 10a was obtained as the unique cycloadduct. The diester 9a was cyclized into the challenging fully substituted diimidazobenzobutane 13 system present in benzosceptrins.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
55. Iron-Catalyzed Sulfur-Promoted Decyanative Redox Condensation of o-Nitrophenols and Arylacetonitriles: an Unprecedented Route to 2-Arylbenzoxazoles
Thanh Binh Nguyen* and Jerome Cheung-Lung
Eur. J. Org. Chem. 2018, 5815.
Nguyen, T. B.; Cheung-Lung, J. Eur. J. Org. Chem. 2018, 5815.
DOI: 10.1002/ejoc.201701607

Abstract. Elemental sulfur in the presence of catalytic amount FeCl2•4H2O was found to be highly efficient to promote decyanative redox condensation reactions of o-nitrophenols 1 with arylacetonitriles 2 to a wide range of 2-arylbenzoxazoles 3. The utility of elemental sulfur was highlighted by its role as cyanide scavenger and external reducing agent.
54. Sulfurative Self-Condensation of Ketones and Elemental Sulfur: a Three-Component Access to Thiophenes Catalyzed by Aniline Acid-Base Conjugate Pairs
Thanh Binh Nguyen* and Jerome Cheung-Lung
Eur. J. Org. Chem. 2018, 5815.
Nguyen, T. B.; Cheung-Lung, J. Eur. J. Org. Chem. 2018, 5815.
DOI: 10.1002/ejoc.201701607

Abstract. Elemental sulfur in the presence of catalytic amount FeCl2•4H2O was found to be highly efficient to promote decyanative redox condensation reactions of o-nitrophenols 1 with arylacetonitriles 2 to a wide range of 2-arylbenzoxazoles 3. The utility of elemental sulfur was highlighted by its role as cyanide scavenger and external reducing agent.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
54. Sulfurative Self-Condensation of Ketones and Elemental Sulfur: a Three-Component Access to Thiophenes Catalyzed by Aniline Acid-Base Conjugate Pairs Thanh Binh Nguyen* and Pascal Retailleau
Green Chem. 2018, 20, 387.
DOI: 10.1039/C7GC03437G
Nguyen, T. B.; Retailleau, P. Green Chem. 2018, 20, 387.
DOI: 10.1039/C7GC03437G
Nguyen, T. B.; Retailleau, P. Green Chem. 2018, 20, 387.
Abstract. A sulfurative self-condensation method for constructing thiophenes 2 by reaction between ketones 1 with elemental sulfur is reported. The reaction, which is catalyzed by anilines and their salt with strong acids, starts from readily available and inexpensive materials, and releases only water as a by-product.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Thanh Binh Nguyen* and Pascal Retailleau
Org. Lett. 2018, 20, 186.
DOI: 10.1021/acs.orglett.ol7b03547
Nguyen, T. B.; Retailleau, P. Org. Lett. 2018, 20, 186.
Abstract. A new mode for the activation of elemental sulfur is reported. In the presence of both DMSO and a tertiary aliphatic amine (triethylamine or N-methylpiperidine), this element reacts directly with a wide range of 2’-nitrochalcones 1 to provide the corresponding thioaurones 2 in high yields even at rt and in the absence of transition metal catalyst.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Org. Lett. 2018, 20, 186.
DOI: 10.1021/acs.orglett.ol7b03547
Nguyen, T. B.; Retailleau, P. Org. Lett. 2018, 20, 186.
Abstract. A new mode for the activation of elemental sulfur is reported. In the presence of both DMSO and a tertiary aliphatic amine (triethylamine or N-methylpiperidine), this element reacts directly with a wide range of 2’-nitrochalcones 1 to provide the corresponding thioaurones 2 in high yields even at rt and in the absence of transition metal catalyst.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
52. Methyl ketone break-and-rebuild: new strategy for fully α-heterofunctionalization of acetophenones
Thanh Binh Nguyen* and Pascal Retailleau
Green Chem. 2017, 19, 5371.
DOI: 10.1039/C7GC02558K
Nguyen, T. B.; Retailleau, P. Green Chem. 2017, 19, 5371.
Abstract. Willgerodt reaction under iron-catalyzed aerobic conditions was found to be an excellent tool for fully α-heterofunctionalization of acetophenones with sulfur and amines. Via this break-and rebuild strategy, a wide range of thioglyoxamides was synthesized in a four-component, highly atom-, step- and redox- economical manner with water as the only by-product.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
51. Elemental Sulfur as Reaction Medium for the Synthesis of Fused Nitrogen Heterocycles by Oxidative Coupling between Cycloalkanones and Nitrogen NucleophilesThanh Binh Nguyen* and Pascal Retailleau
Green Chem. 2017, 19, 5371.
DOI: 10.1039/C7GC02558K
Nguyen, T. B.; Retailleau, P. Green Chem. 2017, 19, 5371.
Abstract. Willgerodt reaction under iron-catalyzed aerobic conditions was found to be an excellent tool for fully α-heterofunctionalization of acetophenones with sulfur and amines. Via this break-and rebuild strategy, a wide range of thioglyoxamides was synthesized in a four-component, highly atom-, step- and redox- economical manner with water as the only by-product.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
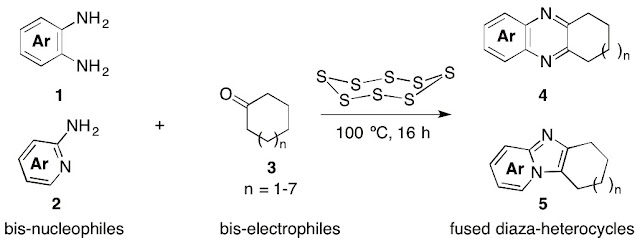
Thanh Binh Nguyen* and Pascal Retailleau
Adv. Synth. Catal. 2017, 359, 3843.
DOI:
Nguyen, T. B.; Retailleau, P. Adv. Synth. Catal. 2017, 359, 3843.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Thanh Binh Nguyen* and Pascal Retailleau
Org. Lett. 2017, 19, 4858.
DOI: 10.1021/acs.orglett.7b02321
Nguyen, T. B.; Retailleau, P. Org. Lett. 2017, 19, 4858.
Nguyen, T. B.; Retailleau, P. Org. Lett. 2017, 19, 4858.
DIPEA was found to be an excellent sulfur activator to promote the reaction of 2-nitrochalcones with elemental sulfur. A wide range of 2-benzoylbenzothiophenes was obtained as a result of a cascade of alkene C═C bond thiolation, aromatic sulfur-denitration.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx48. Elemental Sulfur as Polyvalent Reagent in Redox Condensation with o-Chloronitrobenzenes and Benzaldehydes: Three-Component Access to 2-Arylbenzothiazoles
Le Anh Nguyen, Anh Quoc Ngo, Pascal Retailleau and Thanh Binh Nguyen*
Green Chem. 2017, 19, 4289.
DOI: 10.1039/C7GC01825H
Nguyen, L. A.; Ngo, Q. A.; Retalleau, P.; Nguyen, T. B. Green Chem. 2017, 19, 4289.
Green Chem. 2017, 19, 4289.
DOI: 10.1039/C7GC01825H
Nguyen, L. A.; Ngo, Q. A.; Retalleau, P.; Nguyen, T. B. Green Chem. 2017, 19, 4289.
Abstract. Sulfur was found to be a polyvalent reagent in the multicomponent redox condensation with o-chloronitrobenzenes and benzaldehydes. 2-Arylbenzothiazoles was obtained in moderate to good yields in a highly atom-economical pathway.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
47. Redox-Neutral Access to Sultams from 2-Nitrochalcones and Sulfur with Complete Atom EconomyThanh Binh Nguyen,* Pascal Retailleau
Org. Lett. 2017, 19, 3879.
DOI: 10.1021/acs.orglett.7b01766
Nguyen, T. B.; Retailleau, P. Org. Lett. 2017, 19, 3879.
Abstract. A catalyst-free, redox-neutral, and completely atom-economical synthesis of sultams by simply heating 2-nitrochalcones with elemental sulfur in 3-picoline or N-methylmorpholine is described. The S–N, C–S, and S═O bonds of the sulfonamide are efficiently formed between the nitrogen atom of the 2-nitro group and the α-carbon of the chalcones and elemental sulfur with the migration of two oxygen atoms from the 2-nitro group to the sulfur atom.
www.organic-chemistry.org/
www.organic-chemistry.org/
Thanh Binh Nguyen,* Pascal Retailleau
Org. Lett. 2017, 19, 3887.
DOI: 10.1021/acs.orglett.7b01775Nguyen, T. B.; Retailleau, P. Org. Lett. 2017, 19, 3887.
Abstract. In the presence of N-methylpiperidine, elemental sulfur was found to act as excellent oxidant in promoting oxidative rearranging coupling between o-aminophenols and ketones. A wide range of 2-alkylbenzoxazoles was obtained under mild conditions.
www.organic-chemistry.org/
Org. Lett. 2017, 19, 3887.
www.organic-chemistry.org/
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
45. Recent Advances in Organic Reactions Involving Elemental Sulfur
Thanh Binh NguyenAdv. Synth. Catal. 2017, 359, 1066-1130.
DOI: 10.1002/adsc.201601329
Nguyen, T. B. Adv. Synth. Catal. 2017, 359, 1066-1130.
Abstract. Elemental sulfur has been known since Antiquity and found
widespread applications in the preparation of black gunpowder, the synthesis of
sulfuric acid as well as other sulfur-containing compounds, and vulcanization
of rubber. Over the last several years, we come to better understand its
properties and discover more applications of elemental sulfur in synthetic
organic chemistry. This review summarizes the advances from 2000 in the construction
of organic molecules using elemental sulfur via sulfuration, oxidation,
reduction and redox condensation processes.
1 Introduction
2 Sulfur as Building Block – Sulfuration Reactions
2.1 Sulfuration Reactions with One Substrate
2.2 Sulfuration Reactions with More than One
Substrate
3 Sulfur as Oxidant
4 Sulfur as Reductant
5 Sulfur as Catalyst
5.1 Sulfur as the Only Catalyst
5.2 Iron–Sulfur Catalyst for Redox Condensation
Reactions of the Nitro Group
6 Conclusion
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
44. Elemental sulfur and molecular iodine as efficient tools for carbon-nitrogen bond formation via redox reactions
Thanh Binh Nguyen
Asian J. Org. Chem. 2017, 6, 477-491.
Asian J. Org. Chem. 2017, 6, 477-491.
AsianJOC Readers’ Choice 2019
Abstract. Carbon-nitrogen bonds are ubiquitous in organic chemistry. This review
will analyze some recent advances in the redox reactions leading to
these important linkages involving elemental sulfur and molecular iodine
as excellent reagents/catalysts.
43. Remarkably high homoselectivity in [2 + 2] photodimerization of trans-cinnamic acids in multicomponent systems
Thanh Binh Nguyen* and Ali Al-Mourabit
Photochem. Photobiol. Sci. 2016, 15, 1115-1119.
Nguyen, T. B.; Almourabit, A. Photochem. Photobiol. Sci. 2016, 15, 1115-1119.
DOI: 10.1039/C6PP00201C
Thanh Binh Nguyen* and Ali Al-Mourabit
Photochem. Photobiol. Sci. 2016, 15, 1115-1119.
Nguyen, T. B.; Almourabit, A. Photochem. Photobiol. Sci. 2016, 15, 1115-1119.
DOI: 10.1039/C6PP00201C
Abstract. [2 + 2] homoadducts were exclusively obtained with total regio- and stereo-selectivities when a suspension of several solid photoactive trans-cinnamic acids in cyclohexane was stirred and irradiated.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
42. Molecular Iodine-Catalyzed Aerobic α,β-Diamination of Cyclohexanones with 2-Aminopyrimidine and 2-Aminopyridines
Org. Lett. 2016, 18, 2177.
DOI: 10.1021/acs.orglett.6b00823
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Org. Lett. 2016, 18, 2177.
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Org. Lett. 2016, 18, 2177.
Abstract. Molecular iodine is shown to be an excellent catalyst for aerobic
oxidative α,β-diamination of cyclohexanones with
2-aminopyrimidine/2-aminopyridines. This α,β-C–H functionalization is
remarkable for its simplicity in both substrates and conditions,
involving one and a half oxygen molecules and releasing three water
molecules as the only byproduct. In addition, the functionalized
products including protected 2-aminoimidazoles introduced without
aromatization can serve as useful building blocks for natural product
synthesis and medicinal chemistry.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
41. Formic Acid as a Sustainable and Complementary Reductant: Approach to Fused Benzimidazoles by Molecular Iodine-Catalyzed Reductive Redox Cyclization of o-Nitro-t-Anilines
Thanh Binh Nguyen, Ludmila Ermolenko and Ali Al Mourabit
Green Chem. 2016, 18, 2966.
DOI: 10.1039/C6GC00902F
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Green Chem. 2016, 18, 2966.
Abstract. Molecular iodine was found
to be an excellent catalyst for reductive redox cyclization of
o-nitro-t-anilines 1 into fused tricyclic or 1,2-disubtituted
benzimidazoles 2. A range of functions such as halides (F, Cl, Br),
methoxy, ester, trifluoromethyl, cyano, pyridine and even nitro groups
were tolerated using formic acid as a clean, safe, user-friendly and
complementary reductant. When iodine was used in stoichiometric amount
(50 mol %), the methodology allowed direct synthesis of benzimidazole
hydroiodides 2.HI in high yields by simple precipitation from reaction
mixture.Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Green Chem. 2016, 18, 2966.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
40. Redox condensation of o-Halonitrobenzene with 1,2,3,4-Tetrahydroisoquinoline: Involvement of an Unexpected Auto-catalyzed Redox Cascade
Abstract. A practical synthesis of fused benzimidazoles 5 has been developed by
simply heating o-halonitrobenzenes 1 with tetrahydroisoquinolines 2. In
this transformation, 2 played multiple roles: building block, base and
double hydride donor in a cascade of uncatalyzed aromatic substitution,
reduction of the nitro group, oxidation of the α-methylene group and
condensation.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
39. Vacuum Desiccator as a Simple, Robust, and Inexpensive NMR Tube Cleaner
Abstract. A simple, robust, and inexpensive apparatus for cleaning several NMR
tubes is easily fit up and used from readily available glassware,
including a beaker, a desiccator, and a rotary evaporator vacuum pump.
A Spiffy Way To Clean NMR Tubes
Lab Techniques: A simple vacuum setup and a little solvent offer a fast, inexpensive way to clean many glass tubes at once
By
Life in
a chemistry lab isn’t always fun and games—chemists must also do their chores, including
cleaning out used NMR tubes. Although commercial devices are available for the
task, they are expensive glassware and typically only clean one tube or a few at
a time. To remedy that problem, synthetic organic chemist Thanh Binh Nguyen of
the Institut de Chimie des Substances Naturelles - CNRS has devised an NMR tube cleaning
system that can handle dozens of tubes at once and only requires a small amount
of solvent and equipment already at hand in most labs (Org. Process Res. Dev. 2016, DOI: 10.1021/acs.oprd.6b00001). First, Binh empties NMR tubes and places them upside down in a beaker containing
solvent or cleaning solution. Binh then places the beaker in a vacuum
desiccator, which he evacuates and vents with air several times. The liquid rises
and falls with each vacuum cycle, cleaning out the tubes. A final rinse with
fresh acetone completes the cleaning. Binh says he came up with the idea when
his research funds were short. “I had to optimize everything—time, chemicals,
human power—and here is one of my solutions.” Early responses to the OPR&D paper
on Twitter were mixed: Some commenters questioned publishing the work in an industrial
process chemistry journal, noting that industrial chemists often consider NMR
tubes as a one-time consumable and toss them out. Plus cleaning them creates
more lab waste. But most admit it’s a clever idea.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
38. Elements as Direct Feedstocks for Organic Synthesis: Fe/I2/O2 for Diamination of 2-Cyclohexenones with 2-Aminopyrimidine and 2-Aminopyridines
Thanh Binh Nguyen, Mathilde Corbin, Pascal Retailleau, Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2015, 17, 4956.
DOI: 10.1021/acs.orglett.5b02340
Nguyen, T. B.; Corbin, M.; Retailleau, P.; Ermolenko, L.; Almourabit, A. Org. Lett. 2015, 17, 4956.
Nguyen, T. B.; Corbin, M.; Retailleau, P.; Ermolenko, L.; Almourabit, A. Org. Lett. 2015, 17, 4956.
Abstract. Elements as feedstocks for organic synthesis, the trio of metallic iron,
molecular iodine, and dioxygen, were found to be an excellent tool for
oxidative regioselective diamination of conjugated enones with
2-aminopyrimidine (a guanidine surrogate) and 2-aminopyridines leading
to unaromatized coupled products in moderate to good yields.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
37. Concise Access to 2-Aroylbenzothiazoles by Redox Condensation Reaction between o-Halonitrobenzenes, Acetophenones, and Elemental Sulfur
Thanh Binh Nguyen, Karine Pasturaud, Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2015, 17, 2562-2565.
DOI: 10.1021/acs.orglett.5b01182
Nguyen, T. B.; Pasturaud, K.; Ermolenko, L.; Almourabit, A. Org. Lett. 2015, 17, 2562.
Angew. Chem. Int. Ed. 2014, 53, 13808.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Nguyen, T. B.; Pasturaud, K.; Ermolenko, L.; Almourabit, A. Org. Lett. 2015, 17, 2562.
Abstract. A wide range of 2-aroylbenzothiazoles 3 including some pharmacologically relevant derivatives can be obtained in high yields by simply heating o-halonitrobenzenes 1, acetophenones 2, elemental sulfur, and N-methylmorpholine.
This three-component nitro methyl coupling was found to occur in an
excellent atom-, step-, and redox-efficient manner in which elemental
sulfur played the role of nucleophile building block and redox
moderating agent to fulfill electronic requirements of the global
reaction.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
36. Sodium Sulfide: A Sustainable Solution for Unbalanced Redox Condensation Reaction between o-Nitroanilines and Alcohols Catalyzed by an Iron–Sulfur System
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Elemental Sulfur Disproportionation in the Redox Condensation Reaction between o-Halonitrobenzenes and Benzylamines
Thanh Binh Nguyen, Pascal Retailleau, Ludmila Ermolenko, and Ali Al-Mourabit
Angew. Chem. Int. Ed. 2014, 53, 13808.
Angew. Chem. Int. Ed. 2014, 53, 13808.
Abstract. The disproportionation of elemental sulfur at moderate temperatures is investigated in the redox condensation involving o-halonitrobenzenes 1 and benzylamines 2.
As a redox moderator, elemental sulfur plays the dual role of both
electron donor and acceptor, generating its lowest and highest oxidation
states: S−2 (sulfide equivalent) in benzothiazole 3 and S+6 (sulfate equivalent) in sulfamate 4,
and filling the electron gap of the global redox condensation process.
Along with this process, a cascade of reactions of reduction of the
nitro group of 1, oxidation of the aminomethyl group of 2, metal-free aromatic halogen substitution, and condensation finally led to 2-arylbenzothiazoles 3.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
34. Three-Component Reaction between Isocyanides, Aliphatic Amines and Elemental Sulfur: Preparation of Thioureas under Mild Conditions with Complete Atom Economy
Invited Feature Article
Nguyen, T. B.; Ermolenko, L.; Almourabit, A.
Nguyen, T. B.; Ermolenko, L.; Almourabit, A.
The reaction of isocyanides with aliphatic amines in the presence of
elemental sulfur was found to proceed efficiently at, or near, room
temperature to produce thioureas in excellent yields and with complete
atom economy.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
33. Fe/S-catalyzed decarboxylative redox condensation of arylacetic acids with nitroarenes
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
33. Fe/S-catalyzed decarboxylative redox condensation of arylacetic acids with nitroarenes
Thanh Binh Nguyen,* Ludmila Ermolenko, Mathilde Corbin and Ali Al-Mourabit*
Org. Chem. Front. 2014, 1, 1157.
DOI: 10.1039/C4QO00221K
Nguyen, T. B.; Ermolenko, L.; Corbin, M.; Almourabit, A. Org. Chem. Front. 2014, 1, 1157.
Cover picture: mice's wedding Đông Hồ folk woodcut painting (Tranh khắc gỗ dân gian Đông Hồ)
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Fe/S clusters generated in situ from simple iron salts and sulfur S8
were found to be highly efficient to catalyze the decarboxylative redox
condensation of arylacetic acids with nitroarenes in the presence of N-methylpiperidine
as a basic additive. A wide range of aza-heterocycles was obtained in
an atom-, step-, and redox-economical manner with water and carbon
dioxide as the only by-products.
Cover picture: mice's wedding Đông Hồ folk woodcut painting (Tranh khắc gỗ dân gian Đông Hồ)
30. Reaction of Quinones and Guanidine Derivatives: Simple Access to Bis-2-aminobenzimidazole Moiety of Benzosceptrin and Other Benzazole Motifs
Thanh Binh Nguyen, Minh Quan Tran, Ludmila Ermolenko, Pascal Retailleau, and Ali Al-Mourabit
Org. Lett. 2014, 16, 920.
DOI: 10.1021/ol403672p
Nguyen, T. B.; Tran, M. Q.; Ermolenko, L.; Retailleau, P.; Almourabit, A. Org. Lett. 2014, 16, 920.
Nguyen, T. B.; Tran, M. Q.; Ermolenko, L.; Retailleau, P.; Almourabit, A. Org. Lett. 2014, 16, 920.
Abstract. A new strategy for the synthesis of 2-aminobenzimidazol-6-ols via a
reaction of quinones with guanidine derivatives is reported. Sequential
application of this methodology provided a simple access to the first
benzosceptrin analogue bearing a bis-2-aminoimidazole moiety. A
concomitant addition of two guanidines to the
naphtho[1′,2′:4,5]imidazo[1,2-a]pyrimidine-5,6-dione, which
includes the redox neutral debenzylation and guanidine-assisted cleavage
of the 2-aminopyrimidine part resulted in the synthesis of the free
challenging contiguous bis-2-aminoimidazole moiety of benzosceptrins in
one step.
29. Three-Component Reaction between Alkynes, Elemental Sulfur, and Aliphatic Amines: A General, Straightforward, and Atom Economical Approach to Thioamides
Thanh Binh Nguyen, Minh Quan Tran, Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2014, 16, 310.
DOI: 10.1021/ol403345e
Nguyen, T. B.; Tran, M. Q.; Retailleau, P.; Almourabit, A. Org. Lett. 2014, 16, 310.
Nguyen, T. B.; Tran, M. Q.; Retailleau, P.; Almourabit, A. Org. Lett. 2014, 16, 310.
28. Cobalt- and Iron-Catalyzed Redox Condensation of o-Substituted Nitrobenzenes with Alkylamines: A Step- and Redox-Economical Synthesis of Diazaheterocycles
Thanh Binh Nguyen,* Julie Le Bescont, Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2013, 15, 6218.
DOI: 10.1021/ol403064z
Nguyen, T. B.; Le Bescont, J.; Ermolenko, L.; Almourabit, A. Org. Lett. 2013, 15, 6218.
Nguyen, T. B.; Le Bescont, J.; Ermolenko, L.; Almourabit, A. Org. Lett. 2013, 15, 6218.
27. A Simple and Straightforward Approach to Quinoxalines by Iron/Sulfur-Catalyzed Redox Condensation of o-Nitroanilines and Phenethylamines
Thanh Binh Nguyen,* Pascal Retailleau, and Ali Al-Mourabit
Org. Lett. 2013, 15, 5238.
DOI: 10.1021/ol402435c
Nguyen, T. B.; Retailleau, P.; Almourabit, A. Org. Lett. 2013, 15, 5238.
Nguyen, T. B.; Retailleau, P.; Almourabit, A. Org. Lett. 2013, 15, 5238.
Abstract. In situ generated iron sulfide from elemental sulfur and ferric
chloride was found to be a highly efficient catalyst for the redox
condensation cascade reaction between o-nitroanilines and
2-arylethylamines. This method constitutes a new atom-, step-, and
redox-economical route to 2-arylquinoxalines.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx26. Nitro-Methyl Redox Coupling: Efficient Approach to 2-Hetarylbenzothiazoles from 2-Halonitroarene, Methylhetarene, and Elemental Sulfur
Thanh Binh Nguyen,* Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2013, 15, 4218.
DOI: 10.1021/ol401944a
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Org. Lett. 2013, 15, 4218.
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Org. Lett. 2013, 15, 4218.
Abstract. A simple, straightforward, and atom economic approach to
2-hetarylbenzothiazoles starting from 2-halonitroarene, methylhetarene,
and elemental sulfur under mild conditions is described. The method is
highlighted by the direct redox nitro-methyl reaction for
carbon–nitrogen bond formation without an added oxidizing or reducing
agent.
Highlighted by Synfacts 2013; 9(11): 1160
DOI: 10.1055/s-0033-1340003 xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
25. Selective Autoxidation of Benzylamines: Application to the Synthesis of Some Nitrogen Heterocycles
Thanh Binh Nguyen,* Ludmila Ermolenko and Ali Al-Mourabit
Green Chem. 2013, 15, 2713.
DOI: 10.1039/C3GC41186A
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Green Chem. 2013, 15, 2713.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx

xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
13. Nguyen, T. B., Wang, Q.; Guéritte, F. Synth. Commun, 2012, 42, 2648.
"Practical Synthesis of N-Aryl-o-Hydroxyaryl Ketimines"
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Abstract. A green and remarkably simple synthesis of nitrogen heterocycles from benzylamines and anilines o-substituted by a cyclizable group has been developed based on selective uncatalyzed autoxidation of benzylamines.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
24. Hydrogen Bond Organocatalysis of Benzotriazole in Transamidation of Carboxamides with Amines
Thanh Binh Nguyen,* Ludmila Ermolenko, Marie-Elise Tran Huu Dau, and Ali Al-Mourabit
Heterocycles, Victor Snieckus's Special issue, Vol. 88, No. 1, 2014, pp. 403-416
DOI: 10.3987/COM-13-S(S)41
Nguyen, T. B.; Ermolenko, L.; Tran Huu Dau, M.; Almourabit, A. Heterocycles, 2014, 88, 403.
FREE PDF
Abstract. A new method of transamidation of carboxamides with amines catalyzed by benzotriazole has been developed.
23. Iron Sulfide Catalyzed Redox/Condensation Cascade Reaction between 2-Amino/Hydroxy Nitrobenzenes and Activated Methyl Groups: A Straightforward Atom Economical Approach to 2-Hetaryl-benzimidazoles and -benzoxazoles
J. Am. Chem. Soc. 2013, 135, 118.
DOI: 10.1021/ja311780a
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. J. Am. Chem. Soc. 2013, 135, 118.
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. J. Am. Chem. Soc. 2013, 135, 118.

xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
22. Benzazoles from Aliphatic Amines and o-Amino/Mercaptan/Hydroxyanilines: Elemental Sulfur as a Highly Efficient and Traceless Oxidizing Agent
Thanh Binh Nguyen,* Ludmila Ermolenko, William A. Dean, and Ali Al-Mourabit
Org. Lett. 2012, 14, 5948.
DOI: 10.1021/ol302856w
Nguyen, T. B.; Ermolenko, L.; Dean, W. A.; Almourabit, A. Org. Lett. 2012, 14, 5948.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Abstract. A convenient room-temperature one-pot procedure for the preparation of benzimidazoles derivatives from N-arylamidines has been developed. The reaction of N-aryl-N'-chloro amidines, generated by the treatment of N-aryl amidines with N-chlorosuccinimide, in presence of sodium hydroxide provides benzimidazoles in good to excellent yields. Nitrogen anion generated in situ from succinimide (by-product of the chlorination step using NCS) and hydroxide anion was found to be highly effective as Brønsted base to promote the cyclization into benzimidazole of N-aryl-N'-chloroamidine.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
17. Chiral Phosphoric Acid Catalyzed Enantioselective Transfer Hydrogenation of ortho-Hydroxybenzophenone N
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Nguyen, T. B.; Ermolenko, L.; Dean, W. A.; Almourabit, A. Org. Lett. 2012, 14, 5948.
Abstract. A novel remarkably simple solvent-free and catalyst-free synthesis of benzazoles from alkylamines and o-hydroxy/amino/mercaptan anilines using elemental sulfur as traceless oxidizing agent has been developed.
Highlighted by
Synfact 2013, 148. DOI: 10.1055/s-0032-1318097
Synthesis of Benzazoles Using S8 as the Oxidizing Agent
and by
http://www.organic-chemistry.org/
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Thanh Binh Nguyen,* Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2012, 14, 4274.
Abstract. An efficient
and selective multicomponent oxidative coupling of two different
aliphatic primary amines into thioamides by elemental sulfur under
solvent-free conditions has been developed.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
20. N-Chlorosuccinimide/sodium hydroxide-mediated synthesis of benzimidazoles from amidines under mild conditions
Thanh Binh Nguyen,* Ludmila Ermolenko, and Ali Al-Mourabit
Heterocycles 2012, 86, Prof. Dr. Ei-ichi Negishi's Special Issue, 555.
DOI: 10.3987/COM-12-S(N)53
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Heterocycles 2012, 86, 555.
DOI: 10.3987/COM-12-S(N)53
Nguyen, T. B.; Ermolenko, L.; Almourabit, A. Heterocycles 2012, 86, 555.
Abstract. A convenient room-temperature one-pot procedure for the preparation of benzimidazoles derivatives from N-arylamidines has been developed. The reaction of N-aryl-N'-chloro amidines, generated by the treatment of N-aryl amidines with N-chlorosuccinimide, in presence of sodium hydroxide provides benzimidazoles in good to excellent yields. Nitrogen anion generated in situ from succinimide (by-product of the chlorination step using NCS) and hydroxide anion was found to be highly effective as Brønsted base to promote the cyclization into benzimidazole of N-aryl-N'-chloroamidine.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
19. Boric Acid: A Highly Efficient Catalyst for Transamidation of Carboxamides with Amines
Thanh Binh Nguyen,* Jonathan Sorres, Minh Quan Tran, Ludmila Ermolenko, and Ali Al-Mourabit
Org. Lett. 2012, 14, 3202.
DOI: 10.1021/ol301308c
Nguyen, T. B.; Sorres, J.; Tran, M. Q.; Ermolenko, L.; Almourabit, A. Org. Lett. 2012, 14, 3202.
Nguyen, T. B.; Sorres, J.; Tran, M. Q.; Ermolenko, L.; Almourabit, A. Org. Lett. 2012, 14, 3202.
A novel method of transamidation of
carboxamides with amines using catalytic amounts of readily available
boric acid under solvent-free conditions has been developed. The scope
of the methodology has been demonstrated with (i) primary, secondary,
and tertiary amides and phthalimide and (ii) aliphatic, aromatic,
cyclic, acyclic, primary, and secondary amines.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
18. Synthesis, Biological Evaluation, and Molecular Modeling of Natural and Unnatural Flavonoidal Alkaloids, Inhibitors of Kinases
Thanh Binh Nguyen, Olivier Lozach, Georgiana Surpateanu, Qian Wang, Pascal Retailleau, Bogdan I. Iorga, Laurent Meijer, and Françoise Guéritte
J. Med. Chem. 2012, 55, 2811.
DOI: 10.1021/jm201727w
Abstract. The screening of the ICSN chemical
library on various disease-relevant protein kinases led to the
identification of natural flavonoidal alkaloids of unknown configuration
as potent inhibitors of the CDK1 and CDK5 kinases. We thus developed an
efficient and modular synthetic strategy for their preparation and that
of analogues in order to determine the absolute configuration of the
active natural flavonoidal alkaloids and to provide further insights on
the structure–activity relationships in this series. The structural
determinants of the interaction between some flavonoidal alkaloids with
specific kinases were also evaluated using molecular modeling.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
17. Chiral Phosphoric Acid Catalyzed Enantioselective Transfer Hydrogenation of ortho-Hydroxybenzophenone N![[BOND]](https://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H Ketimines and Applications
H Ketimines and Applications
Thanh Binh Nguyen, Qian Wang, and Françoise Guéritte
Chem. Eur. J. 2011, 17, 9576
Abstract. Unprotected synthesis: The first enantioselective chiral phosphoric acid catalyzed transfer hydrogenation of unprotected ortho-hydroxybenzophenone N![[BOND]](https://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H imines by using a Hantzsch ester as the hydrogen source afforded the corresponding chiral N,O-unprotected ortho-hydroxydiarylmethylamines in high yields with excellent enantioselectivities.
H imines by using a Hantzsch ester as the hydrogen source afforded the corresponding chiral N,O-unprotected ortho-hydroxydiarylmethylamines in high yields with excellent enantioselectivities.
![[BOND]](https://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) H imines by using a Hantzsch ester as the hydrogen source afforded the corresponding chiral N,O-unprotected ortho-hydroxydiarylmethylamines in high yields with excellent enantioselectivities.
H imines by using a Hantzsch ester as the hydrogen source afforded the corresponding chiral N,O-unprotected ortho-hydroxydiarylmethylamines in high yields with excellent enantioselectivities.xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
16. Phosphoric Acid-Catalyzed Enantioselective Transfer Hydrogenation of N-Aryl-ortho-Hydroxybenzophenone Ketimines
Thanh Binh Nguyen, Qian Wang, and Françoise Guéritte
Adv. Synth. Catal. 2011, 353, 257.
10.1002/adsc.201000754
Abstract. The first enantioselective chiral phosphoric acid-catalyzed transfer hydrogenation of N-aryl-ortho-hydroxybenzophenone ketimines using a Hantzsch ester as hydrogen source was developed to afford, after removal of the N-aryl group, the corresponding chiral diarylmethylamines in high yields and enantioselectivities.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
15. Chiral Phosphoric Acid-Catalyzed Enantioselective Transfer Hydrogenation of ortho-Hydroxyaryl Alkyl N−H Ketimines
Thanh Binh Nguyen, Hadjira Bousserouel, Qian Wang, and Françoise Guéritte
Org. Lett., 2010, 12, 4705-4707
DOI: 10.1021/ol102043x
Abstract. The first enantioselective chiral phosphoric acid-catalyzed transfer hydrogenation of unprotected ortho-hydroxyaryl alkyl N−H ketimines using Hantszch di-tert-butyl ester as a reductant is reported. A variety of ortho-hydroxybenzylamines were obtained in good to excellent yields and enantiomeric excesses.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
14. An Efficient One-Step Synthesis of Piperidin-2-yl and Pyrrolidin-2-yl Flavonoid Alkaloids through Phenolic Mannich Reactions
Thanh Binh Nguyen, Qian Wang, and Françoise Guéritte
Eur. J. Org. Chem. 2011, 7076.
An efficient one-step synthesis of piperidin-2-yl
and pyrrolidin-2-yl flavonoid alkaloids was achieved in good to
excellent yields by a highly regioselective phenolic Mannich reaction of
chrysin with cyclic imines or iminium salts. Performing the reaction in
a mixture of H2O/THF in the absence of an external reagent
afforded the C-6 alkylated product exclusively. The same reaction in
water in the presence of NaOH provided the C-8 alkylated flavonoid as a
major product. The reaction has been successfully extended to simple
phenols.
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
13. Nguyen, T. B., Wang, Q.; Guéritte, F. Synth. Commun, 2012, 42, 2648.
"Practical Synthesis of N-Aryl-o-Hydroxyaryl Ketimines"
xxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxxx
Publications prior to postdoc:
12. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. Org. Prep. Proc. Int. 2012, 44, 1.
"1,3-Dipolar Cycloadditions of Nitrones to Hetero-substituted Alkenes Part 2: Sila-, Thia-, Phospha- and Halo-substituted Alkenes"
"1,3-Dipolar Cycloadditions of Nitrones to Hetero-substituted Alkenes Part 2: Sila-, Thia-, Phospha- and Halo-substituted Alkenes"
11. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. Org. Prep. Proc. Int. 2010, 42, 387.
“1,3-Dipolar Cycloadditions of Nitrones to Heterosubsituted Alkenes – Part 1: Oxa and Aza-
Substituted Alkenes”
“1,3-Dipolar Cycloadditions of Nitrones to Heterosubsituted Alkenes – Part 1: Oxa and Aza-
Substituted Alkenes”
10. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. J. Org. Chem. 2010, 75, 611.
“Access to alpha-Substituted Amino Acid Derivatives via 1,3-Dipolar Cycloaddition of alpha-Amino Ester Derived Nitrones”
“Access to alpha-Substituted Amino Acid Derivatives via 1,3-Dipolar Cycloaddition of alpha-Amino Ester Derived Nitrones”
9. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. Synthesis 2009, 3174.
“A Large-Scale Low-Cost Preparation of N-Benzylhydroxylamine Hydrochloride”
“A Large-Scale Low-Cost Preparation of N-Benzylhydroxylamine Hydrochloride”
8. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. Synlett. 2009, 15, 2492.
"N-(β,β-Difluorovinyl)oxazolidin-2-ones: First Synthesis and Application in [3+2]- and [4+2]-
Cycloaddition-Type Reactions"
"N-(β,β-Difluorovinyl)oxazolidin-2-ones: First Synthesis and Application in [3+2]- and [4+2]-
Cycloaddition-Type Reactions"
7. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. Org. Lett. 2008, 10, 4493.
“N-Benzyl Aspartate Nitrones: Unprecedented Single-Step Synthesis and [3 + 2] Cycloaddition Reactions with Alkenes”
“N-Benzyl Aspartate Nitrones: Unprecedented Single-Step Synthesis and [3 + 2] Cycloaddition Reactions with Alkenes”
6. Nguyen, T. B.; Vuong, T. M. H.; Martel, A.; Dhal, R.; Dujardin, G. Tetrahedron: Asymmetry 2008, 19, 2084.
“Practical Asymmetric Access to Carboxy-Differentiated Aspartate Derivatives via 1,3-Dipolar Cycloaddition of a Nitrone with (R)-4-Ethyl-N-vinyloxazolidin-2-one”
“Practical Asymmetric Access to Carboxy-Differentiated Aspartate Derivatives via 1,3-Dipolar Cycloaddition of a Nitrone with (R)-4-Ethyl-N-vinyloxazolidin-2-one”
5. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. Synlett 2008, 2041.
“Trimethylsilyl Trifluoromethanesulfonate-Mediated Addition-Cyclization of N-Vinyloxazolidin-2- ones to Nitrones: an Efficient Access to 4-Substituted-5-Aza-isoxazolidines”
“Trimethylsilyl Trifluoromethanesulfonate-Mediated Addition-Cyclization of N-Vinyloxazolidin-2- ones to Nitrones: an Efficient Access to 4-Substituted-5-Aza-isoxazolidines”
4. Nguyen, T. B.; Martel, A.; Dhal, R.; Dujardin, G. J. Org. Chem. 2008, 73, 2621.
“1,3-Dipolar Cycloaddition of N-Substituted Dipolarophiles and Nitrones: Highly Efficient Solvent-Free Reaction”
“1,3-Dipolar Cycloaddition of N-Substituted Dipolarophiles and Nitrones: Highly Efficient Solvent-Free Reaction”
3. Nguyen, T. B.; Gaulon, C.; Chapin, T.; Tardy, S.; Tatibouet, A.; Rollin, P.; Dhal, R.; Martel, A.; Dujardin, G. Synlett 2006, 3255.
“New N-Substituted Dipolarophiles in 1,3-Dipolar Cycloaddition of Nitrones”
“New N-Substituted Dipolarophiles in 1,3-Dipolar Cycloaddition of Nitrones”
2. Nguyen, T. B.; Castanet, A. S.; Nguyen, T. H.; Nguyen, K. P. P.; Bardeau, J. F.; Gibaud, A.; Mortier, J. Tetrahedron 2006, 62, 647.
“Synthesis of Model Long-Chain ω-Alkenyltrichlorosilanes and Triethoxysilanes for the Formation of Self-Assembled Monolayers”
“Synthesis of Model Long-Chain ω-Alkenyltrichlorosilanes and Triethoxysilanes for the Formation of Self-Assembled Monolayers”
1. Nguyen K. P. P.; Ton, T. Q.; Nguyen, T. B.; Jaureguiberry, G. Vietnamese J. Chem. 2004, 42, 366.
"Some Factors Which Could Induce the Imine – Enamine Tautomerism of Some Schiff Base Adducts of (±)-Gossypol”
"Some Factors Which Could Induce the Imine – Enamine Tautomerism of Some Schiff Base Adducts of (±)-Gossypol”
















































































